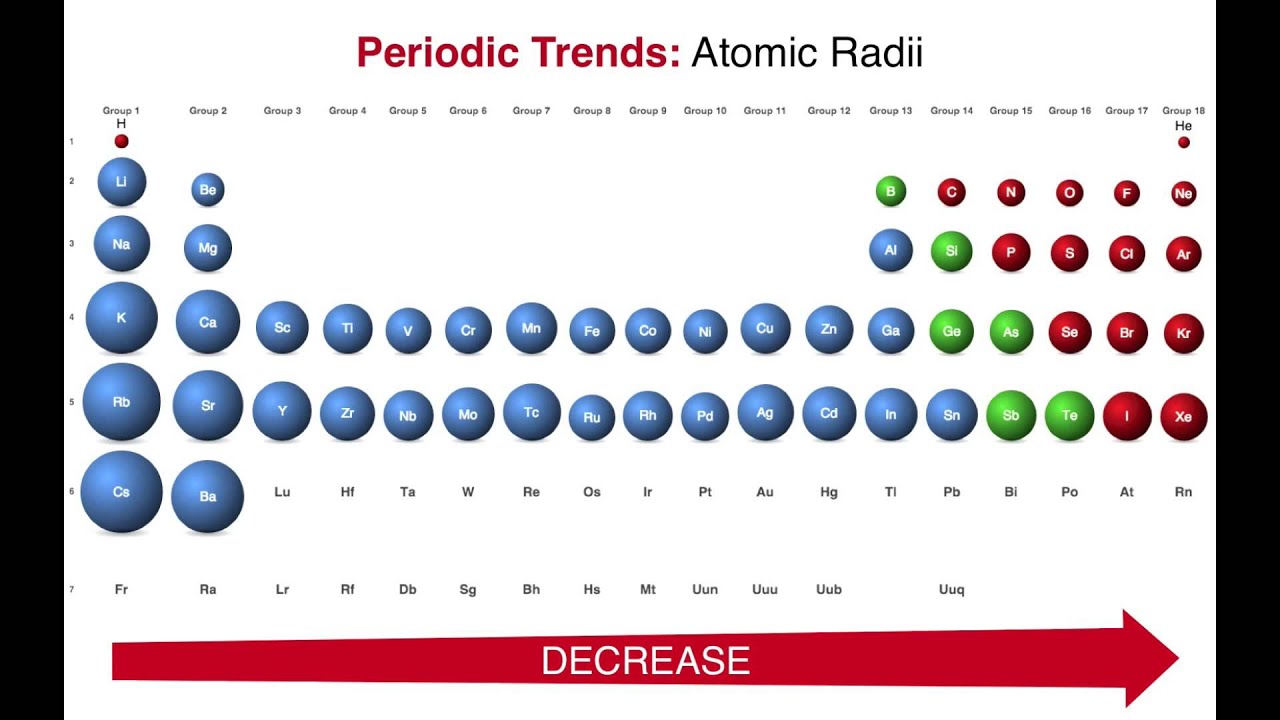
 There are no sharp boundaries for the atomic orbital. The precise measurement of the size of the atom is difficult because of the following reason. Watch The recommended video to better understand atomic radius trends in periodic table The average distance between the nucleus of the atom and its outermost shell provided that the atom is assumed to be spherical is called the atomic radius. Ionization energy or ionization potential.We will discuss the following periodic properties in the modern periodic table. Elements are placed in groups and periods on the basis of similar properties and systematic variation of properties. In the Modern Periodic Table, the elements are arranged in the ascending order of their atomic number. It is of worth importance to know about The variation of different physical and chemical properties in the modern periodic table including groups and periods is known as the periodicity of properties. Periodic trends in the modern periodic table are the basics of chemistry. Trend of melting and boiling points in group.Variation of melting and boiling point in a group.Graphical explanation melting and boiling point in periods.Trend of melting and boiling point in periods. 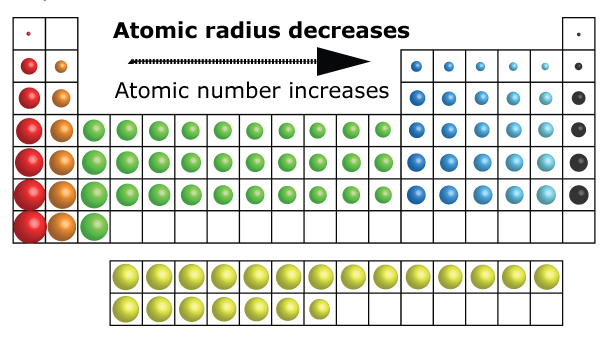
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
